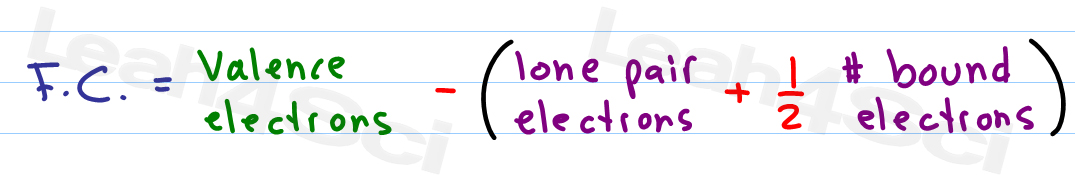
However, the net formal charge in all the resonance structures remains the same. Depending on the compound, the shifting of electrons may cause a change in the atoms’ formal charges. 
These equivalent structures are known as resonance structures. This phenomenon of shifting electrons among the atoms is known as resonance. Formal chargeH 1 (1/2) × 2 0 0 This applies to each hydrogen. The free electrons can move throughout the compound and are said to be delocalized. The difference among these structures is the position of the electrons and bond formation. Sometimes more than one Lewis structure can be drawn to represent the same compound. For formal catering, catering for a buffet or food stations costs 30-150 per person. Dots represent paired electrons, and dash lines show bonded electrons. 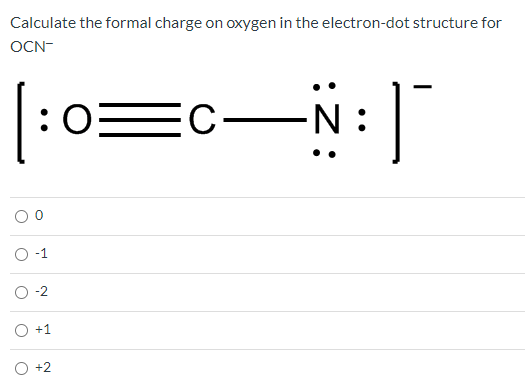
It shows how electrons are positioned around the atoms of the compound. A Lewis dot structure is a straightforward representation of valence shell electrons in an atom, ion, or molecule. In order to calculate the formal charge of a molecule or an ion, one has to know about Lewis structure and resonance. If the compound is neutral, the net formal charge is zero. Use our mortgage calculators to get an idea of how much you could borrow, compare monthly rates and payments, calculate the equity in your property or. The sum of formal charges of all the atoms is equal to the compound’s actual charge. Formal charge is considered to be the charge present in one atom by considering all the bonds to be 100 covalent. This assumption excludes the electronegativity difference between the atoms. How to Calculate the Formal Charge Ī formal charge is a charge assigned to an atom in a molecule, assuming that all electrons in the chemical bonds are shared equally between the atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
